- Home
- Blog
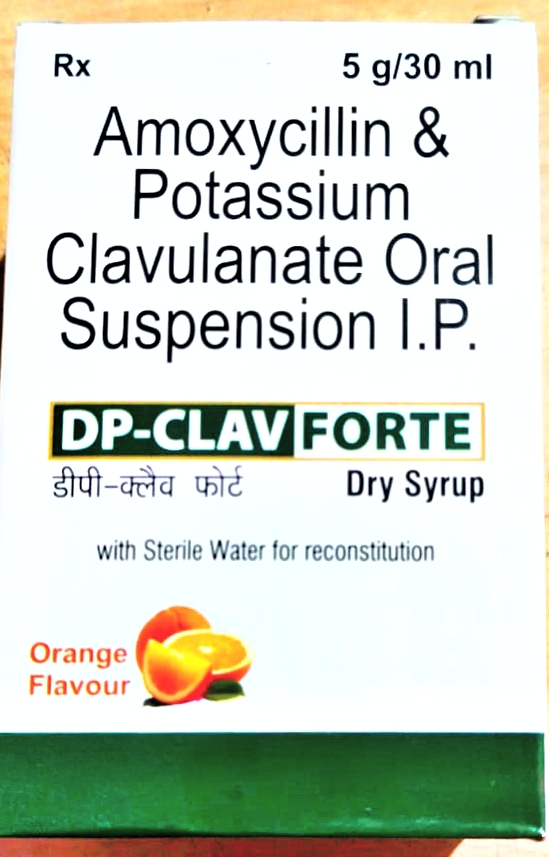
DP-CLAV FORT
Composition
Each 5ml (After reconstitution) contain
Amoxicillin Dehydrator I.P.
Eq to Anhydrous Amoxicillin 400mg
Potassium Clavulanate Diluted I.P.
Eq. to Clavulanic Acid 57mg
Excipients q.s.
Colour: Sunset Yellow FCF
Dosage: As directed by the Physician
Storage: Keep in a dry place at a temperature
not exceeding 300C. Protect from light.
Keep the medicine out of reach of children.
Directions for reconstitution :
- Shake Bottle to loosen Powder.
- Twist and open the vial of sterile water given with the pack.
- Slowly add sterile water upto ring mark on the bottle.
- Put the cap and shake rhe bottle vigorously.
- Adjust the volume upto the ring mark by adding more sterile water, if necessary & shake again, discard the excess water
- Store the reconstituted suspension in the refrigerator. Content to be consumed within 7 days, any extra portion left to be thrown away.
Schedule H prescription drug-caution: Not to be sold by
retail without the prescription of a registered medical practitioner
DO NOT FREEZE
SHAKE WELL RECONSTITUTED
SUSPENSION BEFORE USE
Mfg. Lic. Noi. : 40/UA/SC/P-2008
For manufacturing details of
sterile water for reconstitution refer ampule inside.
Ampule not to be sold separately.
Manufactured in India for :
SATAY SAI REMEDIES & DRUGS (SSRD)
MP 1/35 HB Colony k. Giri-751030
Costumer Care No- 0720575694
Mfg. b y : Rivera Formulation Pvt. Ltd.
ISO 9001 : 2015 &GMP Certified Co.
Plot No. 8, Sector-6A< I.I.E., SIDCUL,
Haridwar-249 403 (U.K.)